Share:
Telomeres are the protective caps on your chromosomes that shorten with every cell division. Understanding what accelerates and what protects telomere length could be the key to slowing biological aging.
# Telomeres and Aging: How to Protect Your Cellular Clock
Imagine your chromosomes as shoelaces. At the end of every shoelace is a small plastic cap — an aglet — that prevents the lace from fraying. Now imagine that every time you tie your shoes, that cap gets a little shorter. Eventually, when the cap is gone, the shoelace begins to unravel.
This is, in essence, what happens to your chromosomes over the course of your lifetime. The protective caps are called **telomeres** — repetitive DNA sequences (TTAGGG in humans) that sit at the ends of chromosomes and shield them from degradation and fusion. And like those aglets, they shorten with every cell division.
Telomere biology has emerged as one of the most compelling frameworks for understanding biological aging, and the 2009 Nobel Prize in Physiology or Medicine was awarded to Elizabeth Blackburn, Carol Greider, and Jack Szostak for their discovery of how chromosomes are protected by telomeres and the enzyme telomerase.
## The Biology of Telomere Shortening
Every time a cell divides, the enzyme that copies DNA — DNA polymerase — cannot fully replicate the very ends of chromosomes. This is known as the **end-replication problem**. As a result, telomeres lose approximately 50–200 base pairs with each cell division.
When telomeres become critically short, cells enter a state called **replicative senescence** — they stop dividing and begin secreting inflammatory molecules. This state, sometimes called the "zombie cell" state, is a key driver of the chronic low-grade inflammation that characterizes aging (a phenomenon researchers call "inflammaging"). Senescent cells accumulate in tissues over time, impairing organ function and contributing to age-related diseases.
The enzyme **telomerase** can counteract telomere shortening by adding new telomeric repeats to chromosome ends. Telomerase is highly active in stem cells and germ cells (sperm and eggs), which is why these cell types can divide many more times than somatic (body) cells. In most adult somatic cells, telomerase activity is low or absent.
## Telomere Length as a Biomarker of Biological Age
Telomere length (TL) has been extensively studied as a biomarker of biological age — a measure of how old your cells are, as distinct from your chronological age. Shorter telomeres are associated with:
| Condition | Evidence Strength |
|---|---|
| Cardiovascular disease | Strong (multiple meta-analyses) |
| Type 2 diabetes | Moderate to strong |
| Alzheimer's disease | Moderate |
| Cancer risk | Complex (short TL increases risk of some cancers) |
| All-cause mortality | Strong |
| Immune dysfunction | Strong |
A landmark study published in *The Lancet* found that individuals with the shortest telomeres had a 3-fold higher risk of heart disease and an 8-fold higher risk of infectious disease mortality compared to those with the longest telomeres. Subsequent meta-analyses involving hundreds of thousands of participants have confirmed that shorter telomere length is a consistent predictor of earlier death.
Importantly, telomere length varies substantially between individuals of the same chronological age — a reflection of both genetic inheritance and lifetime environmental exposures. This variability is what makes telomere biology relevant to longevity interventions.
## What Accelerates Telomere Shortening?
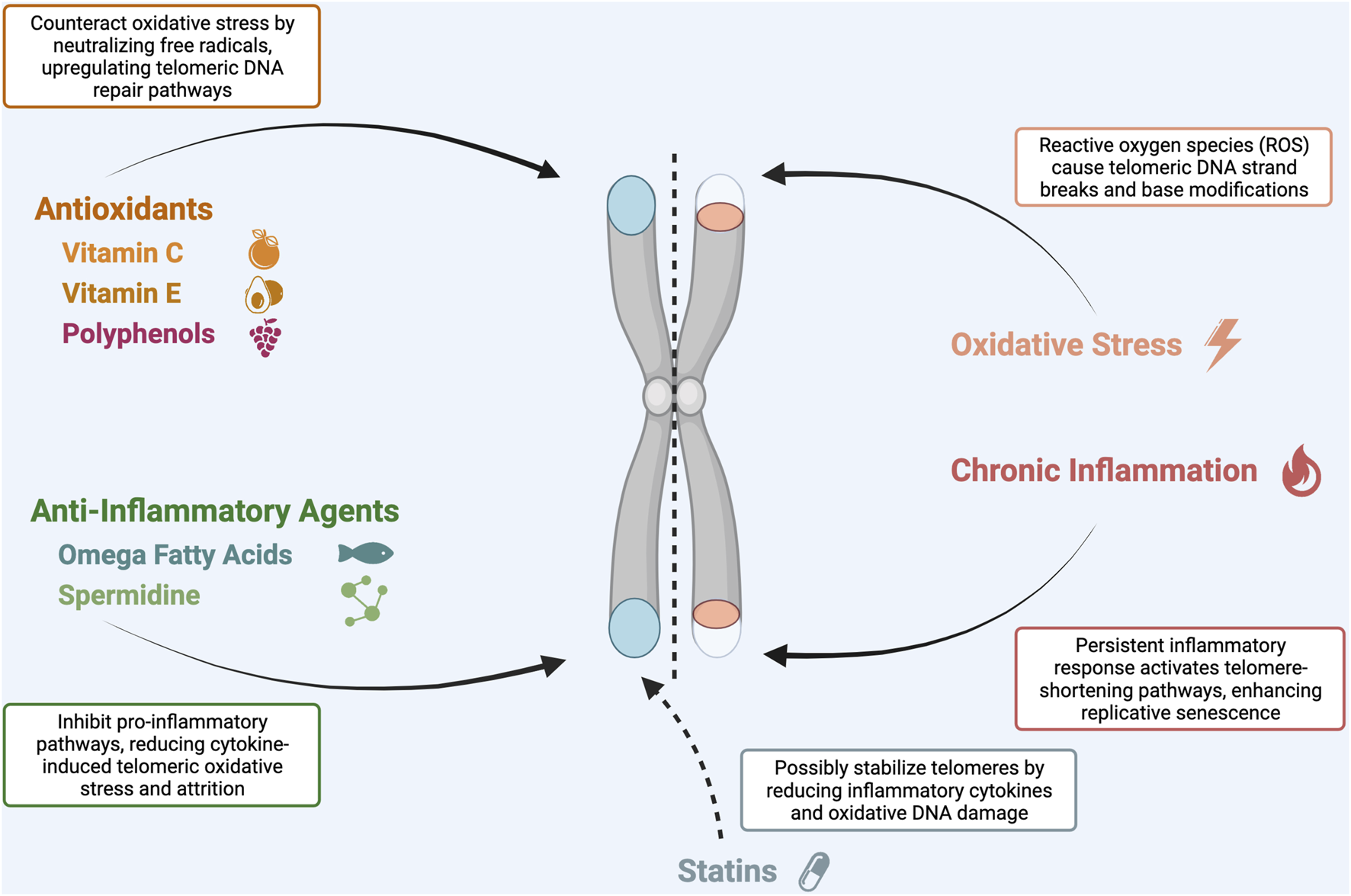
Telomere shortening is not simply a function of cell divisions — it is also driven by oxidative stress and inflammation, which can directly damage telomeric DNA. Several lifestyle and environmental factors have been shown to accelerate telomere attrition:
**Chronic psychological stress** is among the most potent accelerators of telomere shortening. Pioneering research by Elissa Epel and Elizabeth Blackburn demonstrated that mothers of chronically ill children — who experience sustained psychological stress — had significantly shorter telomeres than age-matched controls, equivalent to approximately 10 years of additional cellular aging. The mechanism involves elevated cortisol and oxidative stress.
**Smoking** is strongly associated with shorter telomeres. Each pack-year of smoking is estimated to accelerate telomere shortening by approximately 5 years of biological aging. The oxidative damage from cigarette smoke directly attacks telomeric DNA.
**Obesity and metabolic syndrome** are associated with shorter telomeres, likely through increased oxidative stress, chronic inflammation, and elevated insulin levels. Adipose tissue, particularly visceral fat, is a major source of pro-inflammatory cytokines that damage telomeres.
**Sedentary behavior** independently predicts shorter telomeres, even after controlling for exercise habits. Prolonged sitting is associated with increased inflammatory markers and reduced antioxidant capacity.
**Poor sleep quality** and sleep deprivation are associated with shorter telomeres and reduced telomerase activity, particularly in older adults.
**Processed food consumption**, particularly ultra-processed foods high in refined sugars and industrial seed oils, is associated with shorter telomeres in multiple population studies.
## What Protects and Lengthens Telomeres?
The good news is that telomere length is not fixed — it is modifiable. Several interventions have demonstrated the ability to slow telomere shortening or even increase telomere length:
**Exercise** is the most consistently supported intervention for telomere preservation. Endurance athletes have significantly longer telomeres than sedentary individuals of the same age. A 2017 study in *European Heart Journal* found that endurance exercise and high-intensity interval training (HIIT) increased telomerase activity by 2–3 fold, while resistance training had more modest effects. The minimum effective dose appears to be approximately 150 minutes of moderate aerobic exercise per week.
**Stress reduction practices** have demonstrated measurable effects on telomere length. A landmark study by Dean Ornish and colleagues showed that a comprehensive lifestyle intervention including meditation, yoga, dietary changes, and social support increased telomerase activity by 30% in men with prostate cancer. Mindfulness-based stress reduction (MBSR) programs have also been associated with increased telomerase activity in multiple trials.
**Dietary patterns** matter significantly. The Mediterranean diet — rich in antioxidants, omega-3 fatty acids, and polyphenols — is consistently associated with longer telomeres in epidemiological studies. Specific nutrients with telomere-protective effects include:
- **Omega-3 fatty acids**: A study in *JAMA* found that higher omega-3 levels predicted slower telomere attrition over 5 years
- **Vitamin D**: Deficiency is associated with shorter telomeres; supplementation may help
- **Folate and B vitamins**: Essential for DNA methylation and repair processes that protect telomeres
- **Antioxidants** (vitamins C and E, polyphenols): Reduce oxidative damage to telomeric DNA
**Sleep optimization** is emerging as a critical telomere-protective factor. Studies show that 7–8 hours of quality sleep is associated with longer telomeres, while both short sleep (under 6 hours) and long sleep (over 9 hours) are associated with shorter telomeres.
**Social connection** has a surprisingly robust association with telomere length. Loneliness and social isolation are associated with shorter telomeres, while strong social networks predict longer telomeres — an effect that persists after controlling for other health behaviors.
## Telomerase Activation: The Frontier
Given that telomerase can rebuild telomeres, scientists have long sought ways to safely activate this enzyme in adult somatic cells. The challenge is that telomerase activation also carries cancer risk — many cancer cells achieve immortality by reactivating telomerase.
**TA-65**, a telomerase activator derived from the herb *Astragalus membranaceus*, has been studied in small human trials. Some studies show modest increases in telomere length with TA-65 supplementation, though the evidence is not yet conclusive and the long-term safety profile requires further study.
More promising are approaches that selectively eliminate senescent cells (senolytics) rather than simply extending telomeres. Drugs like dasatinib and quercetin have shown the ability to clear senescent cells in animal models, with early human trials showing improvements in physical function and inflammatory markers.
## The Practical Bottom Line
Telomere biology reinforces a consistent message: the lifestyle factors that protect cellular health — regular vigorous exercise, stress management, quality sleep, a whole-food diet rich in antioxidants, strong social connections, and avoidance of smoking — are the same factors that preserve telomere length and slow biological aging.
You cannot stop telomere shortening entirely, but you have substantial influence over the rate at which it occurs. The choices you make today are quite literally shaping the length of your cellular clock.
Written by
Dr. Marcus Webb
Health & wellness expert contributing evidence-based content to Health Hub.