Share:
Caloric restriction is the most consistently replicated longevity intervention in laboratory history. But does it work in humans, and is it practical? A deep dive into the evidence.
# Caloric Restriction and Longevity: What the Science Actually Says
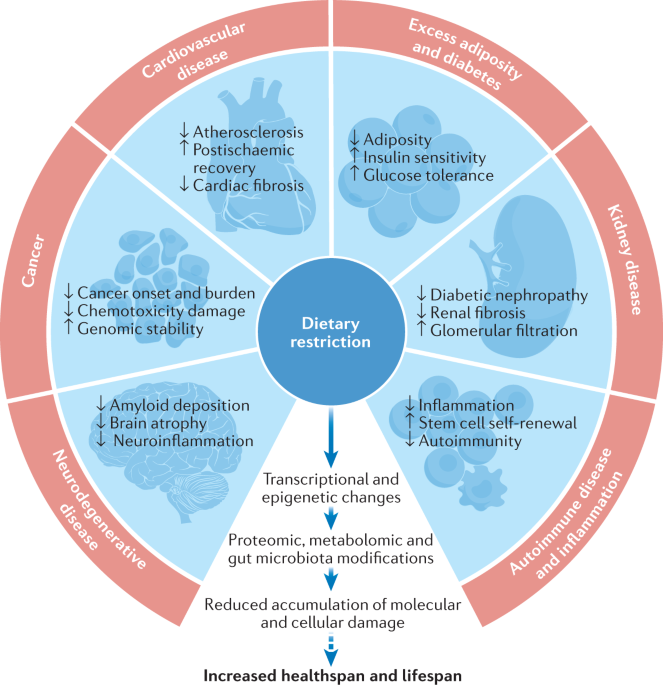
Of all the interventions that scientists have tested for their ability to extend lifespan, one stands out for its consistency across species: caloric restriction (CR). Reducing caloric intake by 20–40% without malnutrition has extended the lifespan of virtually every organism in which it has been tested — from yeast and roundworms to fruit flies, mice, rats, and several species of fish.
The question that has occupied longevity researchers for decades is whether the same holds true for humans — and if so, how to achieve the benefits without the considerable challenges of sustained caloric restriction.
## The History of Caloric Restriction Research
The modern era of caloric restriction research began in 1935, when Clive McCay at Cornell University demonstrated that rats fed a calorie-restricted diet lived nearly twice as long as freely fed controls. This finding was so striking and reproducible that it launched a field of research that continues to generate major discoveries nearly a century later.
In the 1980s and 1990s, researchers at the National Institute on Aging began studying caloric restriction in rhesus monkeys — primates with a much closer evolutionary relationship to humans. The results, published after decades of follow-up, were remarkable: calorie-restricted monkeys showed significantly reduced rates of age-related diseases including diabetes, cardiovascular disease, and cancer, and appeared biologically younger by multiple measures. One study from the University of Wisconsin showed a significant survival advantage for CR monkeys; a parallel study from the NIA showed reduced disease incidence but less clear survival benefit, likely due to differences in diet composition.
## How Caloric Restriction Extends Lifespan: The Mechanisms
Caloric restriction does not extend lifespan through a single mechanism but through a coordinated shift in cellular physiology that favors maintenance and repair over growth and reproduction. The key pathways involved include:
**mTOR inhibition.** mTOR (mechanistic target of rapamycin) is a master regulator of cellular growth that is activated by nutrient availability. When calories are restricted, mTOR activity decreases, which reduces protein synthesis and cell growth while activating autophagy — the cellular recycling process that removes damaged components. mTOR inhibition is one of the most robust longevity pathways identified in model organisms.
**AMPK activation.** AMPK (AMP-activated protein kinase) is the cell's energy sensor. When energy is scarce, AMPK is activated, which promotes fat burning, mitochondrial biogenesis, and autophagy while inhibiting mTOR. AMPK activation mimics many of the effects of caloric restriction.
**Sirtuin activation.** Sirtuins are a family of proteins (SIRT1–SIRT7) that regulate gene expression, DNA repair, and metabolism in response to nutrient availability. Caloric restriction activates sirtuins, particularly SIRT1 and SIRT3, which promote mitochondrial function, reduce oxidative stress, and modulate inflammatory pathways.
**Reduced oxidative stress.** Caloric restriction reduces the production of reactive oxygen species (ROS) by mitochondria, decreasing oxidative damage to DNA, proteins, and lipids — a key driver of cellular aging.
**Hormesis.** Caloric restriction may also work through hormesis — the principle that mild stress triggers adaptive responses that make cells more resilient. The mild metabolic stress of reduced caloric intake activates stress-response pathways that enhance cellular repair and survival.
| Mechanism | Effect on Aging |
|---|---|
| mTOR inhibition | Activates autophagy, reduces cellular growth |
| AMPK activation | Promotes fat oxidation, mitochondrial biogenesis |
| Sirtuin activation | Enhances DNA repair, reduces inflammation |
| Reduced ROS production | Decreases oxidative damage to cellular components |
| Hormesis | Activates stress-response and repair pathways |
## Evidence in Humans: The CALERIE Trial
The most rigorous human study of caloric restriction is the CALERIE (Comprehensive Assessment of Long-term Effects of Reducing Intake of Energy) trial, a randomized controlled trial that enrolled 218 healthy, non-obese adults and assigned them to either a 25% caloric restriction group or an ad libitum (eat as desired) control group for two years.
The results, published in *The Lancet Diabetes & Endocrinology* and other journals, were instructive. Participants in the CR group achieved an average caloric reduction of about 12% (less than the target 25%), resulting in an average weight loss of approximately 7.5 kg. Despite the modest restriction, the CR group showed:
- Significantly reduced cardiometabolic risk factors (blood pressure, LDL cholesterol, triglycerides, insulin resistance)
- Reduced markers of systemic inflammation (C-reactive protein, TNF-alpha)
- Improved quality of life and mood scores
- Reduced metabolic rate (a potential longevity signal, as lower metabolic rate is associated with longer lifespan in some species)
- Preserved lean muscle mass (contrary to earlier concerns)
Importantly, the CALERIE trial also found that caloric restriction was associated with improvements in thyroid hormone levels and body temperature — both markers of a metabolic shift toward longevity-associated physiology.
## Caloric Restriction Mimetics
Given the practical challenges of sustained caloric restriction — hunger, social disruption, potential nutrient deficiencies, and the difficulty of long-term adherence — researchers have invested heavily in identifying compounds that activate CR pathways without requiring food restriction. These are called **caloric restriction mimetics (CRMs)**.
**Rapamycin** is the most potent and well-studied CRM. By directly inhibiting mTOR, rapamycin recapitulates many of the molecular effects of caloric restriction. In mice, rapamycin extends lifespan by 10–25% even when started in late middle age. Human trials are underway, though concerns about immunosuppression at higher doses require careful dose optimization.
**Metformin** activates AMPK and inhibits mTOR, mimicking the metabolic effects of caloric restriction. Epidemiological data consistently show that diabetic patients on metformin have lower rates of cancer, cardiovascular disease, and all-cause mortality than those on other diabetes medications — and in some analyses, lower mortality than non-diabetic controls not taking metformin.
**Resveratrol** activates SIRT1 and has demonstrated longevity effects in yeast, worms, and flies. Results in mammals have been more mixed, and bioavailability in humans is poor with standard formulations. More bioavailable forms (pterostilbene, resveratrol with piperine) are under investigation.
**Spermidine** is a naturally occurring polyamine found in wheat germ, aged cheese, and mushrooms that activates autophagy and has demonstrated lifespan extension in multiple model organisms. A 2018 observational study in humans found that higher dietary spermidine intake was associated with reduced cardiovascular mortality.
**NAD+ precursors** (NMN and NR) support sirtuin activity by replenishing NAD+ levels, which decline with age. Early human trials show improvements in metabolic markers, though long-term longevity data in humans are not yet available.
## Intermittent Fasting as a Practical Alternative
For most people, sustained caloric restriction is not a realistic long-term strategy. Intermittent fasting (IF) — which cycles between periods of eating and fasting rather than continuously restricting calories — has emerged as a more practical approach that activates many of the same pathways.
The most studied IF protocols include:
- **16:8** (16 hours fasting, 8 hours eating window): The most popular and easiest to sustain
- **5:2** (5 days normal eating, 2 days of 500–600 kcal): Studied extensively by Michael Mosley
- **Alternate day fasting**: Alternating between normal eating days and fasting or very low calorie days
- **Prolonged fasting** (24–72 hours, periodically): Maximally activates autophagy and stem cell regeneration
Research suggests that IF produces metabolic benefits comparable to continuous caloric restriction, even when total weekly caloric intake is similar — suggesting that the timing of food intake, not just the quantity, matters for longevity pathways.
## The Practical Bottom Line
The evidence for caloric restriction as a longevity intervention in humans is promising but not definitive. What is clear is that the metabolic pathways activated by caloric restriction — mTOR inhibition, AMPK activation, sirtuin activation, and autophagy induction — are genuine longevity pathways that can be targeted through multiple approaches.
For most people, the most practical strategy is not strict caloric restriction but a combination of: maintaining a healthy body weight, practicing time-restricted eating or intermittent fasting, eating a nutrient-dense diet that avoids ultra-processed foods, and incorporating regular exercise. These approaches activate CR pathways without requiring the sustained hunger and social disruption of continuous caloric restriction — and the evidence suggests they may deliver much of the same benefit.
Written by
James Thornton, RD
Health & wellness expert contributing evidence-based content to Health Hub.