Share:
Senescent cells — cells that have stopped dividing but refuse to die — accumulate throughout the body with age and drive inflammation, tissue dysfunction, and disease. A new class of drugs called senolytics may be able to clear them.
# Senescent Cells and Senolytics: Clearing the Zombie Cells That Drive Aging
In the biology of aging, few concepts have generated as much excitement — and as much therapeutic promise — as cellular senescence. Once considered a relatively obscure cellular phenomenon, senescence has been elevated to a central position in aging biology, and the drugs designed to target senescent cells have become among the most eagerly anticipated interventions in longevity medicine.
The story of senescent cells is, in essence, the story of how the body's own protective mechanisms can become drivers of the very diseases they were designed to prevent.
## What Are Senescent Cells?
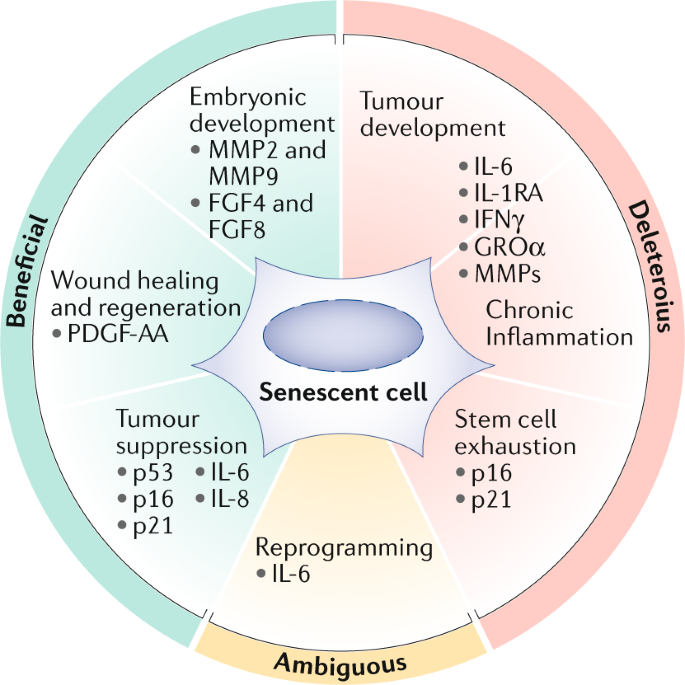
Cellular senescence is a state in which a cell permanently stops dividing in response to stress. First described by Leonard Hayflick in 1961 — who observed that human cells in culture could only divide a finite number of times before entering a non-dividing state (now called the Hayflick limit) — senescence is now understood to be triggered by multiple stimuli: telomere shortening, DNA damage, oncogene activation, oxidative stress, and mitochondrial dysfunction.
Senescence serves important biological functions. In the context of cancer prevention, it acts as a powerful tumor suppressor: when a cell accumulates oncogenic mutations that would otherwise drive uncontrolled proliferation, senescence halts division and prevents malignant transformation. In wound healing, senescent cells transiently appear at injury sites, where they secrete growth factors that promote tissue repair before being cleared by the immune system.
The problem arises when senescent cells are not efficiently cleared. In young, healthy organisms, the immune system — particularly natural killer (NK) cells and macrophages — recognizes and eliminates senescent cells relatively efficiently. With age, immune surveillance declines, and senescent cells begin to accumulate in tissues throughout the body.
## The SASP: Why Senescent Cells Are Toxic
Senescent cells are not merely inert. They actively secrete a complex mixture of pro-inflammatory cytokines, chemokines, growth factors, and proteases collectively known as the **senescence-associated secretory phenotype (SASP)**. The SASP includes molecules such as IL-6, IL-8, TNF-alpha, MMP-3, and VEGF — a cocktail that creates a chronic inflammatory microenvironment in surrounding tissues.
The consequences of SASP accumulation are far-reaching:
| SASP Effect | Disease Consequence |
|---|---|
| Chronic inflammation ("inflammaging") | Cardiovascular disease, neurodegeneration, metabolic syndrome |
| Tissue remodeling via MMPs | Loss of tissue architecture and function |
| Paracrine senescence induction | Spread of senescence to neighboring healthy cells |
| Stem cell niche disruption | Impaired tissue regeneration and repair |
| Immune system dysregulation | Reduced immune surveillance, increased autoimmunity |
| Pro-tumorigenic signaling | Paradoxically promotes cancer progression in established tumors |
This chronic, low-grade inflammation driven by senescent cells — termed "inflammaging" by researcher Claudio Franceschi — is now recognized as a unifying mechanism underlying many age-related diseases, including Alzheimer's disease, atherosclerosis, type 2 diabetes, osteoarthritis, and pulmonary fibrosis.
## The Landmark Mouse Studies
The causal role of senescent cells in aging was definitively established by a series of landmark experiments from the van Deursen laboratory at the Mayo Clinic. In a 2011 *Nature* paper, Baker and colleagues engineered mice to express a "suicide gene" specifically in senescent cells, allowing them to selectively eliminate senescent cells on demand. When these mice were treated to clear their senescent cells, they showed dramatically delayed onset of age-related pathologies — including cataracts, muscle wasting, and fat loss — compared to untreated controls.
In a follow-up 2016 *Nature* paper, the same group showed that clearing senescent cells in already-aged mice extended median lifespan by 25% and improved multiple measures of physical function, including exercise capacity, kidney function, and heart function. Crucially, this benefit was observed even when treatment began in late middle age — suggesting that senolytic interventions might be effective even when started after significant senescent cell accumulation has occurred.
These results were transformative. They provided the first direct causal evidence that senescent cells drive aging phenotypes and that their removal could reverse aspects of aging — not merely slow them.
## Senolytics: The Drugs That Clear Senescent Cells
The discovery that clearing senescent cells could extend healthspan and lifespan in mice immediately prompted the search for drugs that could do the same in humans. These drugs — called **senolytics** — work by targeting the survival mechanisms that allow senescent cells to resist apoptosis (programmed cell death) despite being stressed and dysfunctional.
Senescent cells survive by upregulating anti-apoptotic pathways, particularly those involving BCL-2 family proteins (BCL-2, BCL-XL, BCL-W) and the PI3K/AKT pathway. Senolytics exploit this dependency by inhibiting these survival signals, selectively inducing apoptosis in senescent cells while leaving normal cells unaffected.
The most studied senolytic combination is **dasatinib + quercetin (D+Q)**:
**Dasatinib** is an FDA-approved cancer drug (a BCR-ABL and Src kinase inhibitor) that was identified as a senolytic through a bioinformatics screen. It is particularly effective at clearing senescent fat cell precursors (preadipocytes) and endothelial cells.
**Quercetin** is a flavonoid found naturally in onions, apples, and capers that inhibits PI3K and BCL-2 family proteins. It is particularly effective at clearing senescent human umbilical vein endothelial cells and bone marrow stem cells.
The combination of D+Q is more effective than either drug alone across multiple cell types. In aged mice, D+Q treatment improved physical function, reduced frailty, extended lifespan, and reduced markers of inflammation. In a mouse model of pulmonary fibrosis, D+Q treatment improved lung function and survival.
Other senolytics under investigation include:
- **Navitoclax (ABT-263)**: A potent BCL-2/BCL-XL inhibitor that is highly effective but causes thrombocytopenia (platelet reduction) at therapeutic doses, limiting its clinical use
- **Fisetin**: A flavonoid found in strawberries that has shown senolytic activity in multiple mouse studies and is currently in human trials
- **Piperlongumine**: A natural compound from long pepper with senolytic properties
- **FOXO4-DRI**: A peptide that disrupts the interaction between FOXO4 and p53 in senescent cells, inducing their apoptosis
## Human Clinical Trials: Early Results
The translation of senolytics from mice to humans is still in its early stages, but initial results are encouraging. The first human senolytic trial, published in *EBioMedicine* in 2019, enrolled 14 patients with idiopathic pulmonary fibrosis (IPF) — a fatal lung disease strongly associated with senescent cell accumulation. Patients received intermittent D+Q treatment (3 days on, 4 days off for 3 weeks). Despite the small sample size and lack of a control group, the treated patients showed significant improvements in physical function measures including walking speed, chair-stand test performance, and 6-minute walk distance.
Subsequent trials have examined D+Q in diabetic kidney disease, Alzheimer's disease, osteoporosis, and frailty. A 2021 trial in patients with diabetic kidney disease found that D+Q treatment reduced senescent cell burden (measured by p16 and p21 expression in fat tissue biopsies) and reduced circulating SASP factors. A trial in patients with Alzheimer's disease is currently ongoing.
Importantly, senolytics are typically administered intermittently rather than continuously — a "hit and run" approach that takes advantage of the fact that senescent cells take weeks to months to re-accumulate after clearance. This intermittent dosing strategy minimizes side effects while maintaining efficacy.
## Senostatics: An Alternative Approach
Parallel to senolytics, researchers are developing **senostatics** (also called senomorphics) — compounds that suppress the SASP without killing senescent cells. These include:
- **Rapamycin**: Inhibits mTOR, which is required for SASP production
- **JAK inhibitors** (ruxolitinib, baricitinib): Block the JAK-STAT pathway that drives many SASP components
- **Metformin**: Reduces NF-κB activation and SASP secretion
- **Resveratrol and other SIRT1 activators**: Suppress NF-κB-driven inflammation
Senostatics may be particularly useful in contexts where killing senescent cells is not desirable (such as during wound healing) or where the SASP itself, rather than the senescent cells, is the primary driver of pathology.
## The Road Ahead
Senolytic therapy represents one of the most mechanistically grounded and clinically promising approaches in longevity medicine. The causal evidence from mouse studies is unusually strong, the first human trials are encouraging, and the biological rationale is clear. However, several important questions remain.
The optimal dosing regimen, frequency, and duration of senolytic treatment in humans are not yet established. The long-term safety of repeatedly clearing senescent cells — which serve important functions in wound healing and tumor suppression — requires careful study. And the challenge of measuring senescent cell burden non-invasively in humans (to guide treatment decisions and monitor response) remains a significant technical hurdle.
Nevertheless, the field is advancing rapidly. Multiple senolytic compounds are in clinical trials, biomarker development is progressing, and the first generation of senolytic drugs may reach clinical practice within the next decade. For the first time in the history of medicine, we may be approaching a treatment that targets not a single disease, but a fundamental driver of aging itself.
Written by
Dr. Marcus Webb
Health & wellness expert contributing evidence-based content to Health Hub.