Share:
The 38 trillion microorganisms living in your gut do far more than aid digestion. They regulate your immune system, produce longevity-associated metabolites, and their composition is one of the strongest predictors of healthy aging.
# The Gut Microbiome and Longevity: How Your Trillions of Microbial Allies Shape How Long You Live
The human gut contains approximately 38 trillion microorganisms — bacteria, archaea, fungi, and viruses — that collectively weigh about 1.5 kilograms and encode 150 times more genes than the human genome. For most of medical history, these organisms were viewed primarily as passengers or potential pathogens. The revolution in microbiome science over the last two decades has revealed something far more profound: the gut microbiome is a dynamic, metabolically active organ that profoundly influences human health, disease susceptibility, and — increasingly, the evidence suggests — how long we live.
## The Aging Microbiome: What Changes and Why It Matters
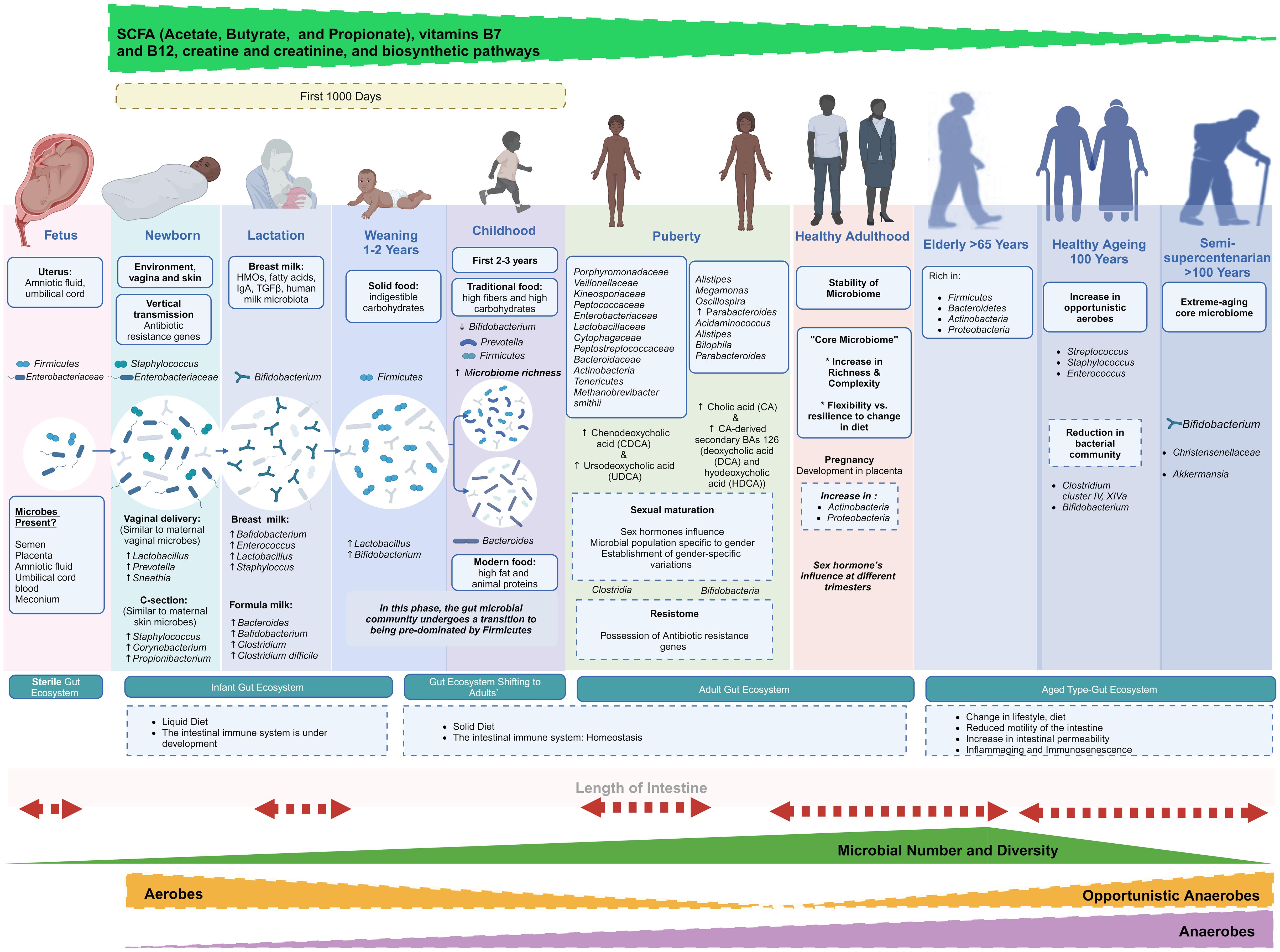
The composition of the gut microbiome changes dramatically across the human lifespan. In infancy, the microbiome is relatively simple and dominated by *Bifidobacterium* species that help digest breast milk. By adulthood, it stabilizes into a complex, individualized ecosystem. With aging, however, the microbiome undergoes characteristic shifts that are associated with declining health:
**Reduced diversity** is the most consistent finding in studies of elderly microbiomes. Microbial diversity — the number of different species present — is a robust predictor of health outcomes. Studies consistently show that older adults with lower microbiome diversity have higher rates of frailty, cognitive decline, inflammation, and all-cause mortality.
**Shifts in dominant taxa** are equally important. Aging is associated with a decline in beneficial bacteria such as *Faecalibacterium prausnitzii*, *Bifidobacterium*, and *Akkermansia muciniphila*, and an increase in pro-inflammatory bacteria such as *Proteobacteria* and *Enterobacteriaceae*. This dysbiosis — an imbalance in microbial composition — is associated with increased intestinal permeability ("leaky gut") and systemic inflammation.
**Reduced production of short-chain fatty acids (SCFAs)** is a critical consequence of these shifts. SCFAs — particularly butyrate, propionate, and acetate — are produced by beneficial bacteria fermenting dietary fiber. Butyrate is the primary energy source for colonocytes (intestinal epithelial cells), maintains intestinal barrier integrity, reduces inflammation, and has been shown to extend lifespan in model organisms. The decline in SCFA-producing bacteria with age is one of the key mechanisms linking microbiome aging to systemic health decline.
## Centenarian Microbiomes: Clues from the Longest-Lived
Some of the most compelling evidence for the microbiome-longevity connection comes from studies of centenarians — individuals who have lived to 100 years or beyond. These studies consistently reveal that centenarians have distinct microbiome profiles that differ markedly from both younger adults and from "ordinary" elderly individuals.
A landmark 2021 study published in *Nature Aging* analyzed the gut microbiomes of 1,575 Chinese individuals aged 20–117, including 160 centenarians. The centenarians had significantly higher microbial diversity and were enriched in bacteria associated with the production of secondary bile acids — a class of metabolites with potent anti-inflammatory and metabolic regulatory properties. Notably, the microbiome profiles of the centenarians were more similar to those of 30-year-olds than to those of typical 80-year-olds.
A Japanese study of supercentenarians (individuals over 110 years old) found that they were enriched in *Christensenellaceae* — a family of bacteria associated with leanness and metabolic health — and in bacteria producing isoallolithocholic acid, a bile acid with potent antimicrobial and immune-regulatory properties.
These findings suggest that maintaining a youthful, diverse microbiome is not merely a consequence of healthy aging but may be a contributing cause of it.
## Key Longevity-Associated Microbial Metabolites
The microbiome influences longevity not just through its composition but through the metabolites it produces. Several classes of microbial metabolites have emerged as particularly important:
| Metabolite Class | Key Examples | Longevity-Relevant Effects |
|---|---|---|
| Short-chain fatty acids | Butyrate, propionate, acetate | Intestinal barrier integrity, anti-inflammation, AMPK activation |
| Secondary bile acids | Isoallolithocholic acid, ursodeoxycholic acid | Immune regulation, antimicrobial activity, metabolic regulation |
| Urolithins | Urolithin A | Mitophagy activation, muscle function, anti-aging in animal models |
| Indoles | Indole-3-propionic acid | Neuroprotection, intestinal barrier integrity, antioxidant |
| Trimethylamine N-oxide (TMAO) | TMAO | Pro-atherogenic (negative — associated with cardiovascular disease) |
**Urolithin A** deserves special mention. Produced by gut bacteria from ellagitannins found in pomegranates, walnuts, and berries, urolithin A activates mitophagy — the selective removal of damaged mitochondria — and has extended lifespan in *C. elegans* and improved muscle function in aged mice. A human clinical trial published in *Nature Metabolism* found that urolithin A supplementation improved mitochondrial function and muscle endurance in older adults. Notably, only about 40% of people have the gut bacteria needed to produce urolithin A from dietary precursors — the rest would need to supplement directly.
**Trimethylamine N-oxide (TMAO)** illustrates the darker side of the microbiome-longevity relationship. TMAO is produced when gut bacteria metabolize choline and carnitine (found in red meat, eggs, and fish) into trimethylamine, which is then converted to TMAO in the liver. High TMAO levels are associated with accelerated atherosclerosis, thrombosis, and cardiovascular mortality. This is one mechanism by which high red meat consumption may increase cardiovascular risk — not through saturated fat alone, but through microbiome-mediated TMAO production.
## The Gut-Brain Axis and Cognitive Aging
The microbiome's influence on longevity extends to the brain through the **gut-brain axis** — a bidirectional communication network involving the vagus nerve, immune signaling, and microbial metabolites. Emerging evidence suggests that gut dysbiosis contributes to neuroinflammation and cognitive aging.
Studies in germ-free mice (which lack a microbiome) show accelerated brain aging and cognitive decline compared to conventionally colonized mice. Transplanting the microbiome of young mice into old mice has been shown to improve cognitive function and reduce neuroinflammation in the recipients — a striking demonstration of the microbiome's causal role in brain aging.
In humans, multiple studies have found associations between gut microbiome composition and cognitive performance in older adults. The *Lactobacillus* and *Bifidobacterium* genera — which decline with age — are particularly associated with cognitive health, likely through their production of GABA, serotonin precursors, and anti-inflammatory metabolites.
Alzheimer's disease patients show consistent microbiome alterations compared to cognitively healthy controls, including reduced *Firmicutes* and increased *Bacteroidetes*, with higher levels of pro-inflammatory bacteria. Whether these changes are cause or consequence of neurodegeneration remains an active area of investigation, but the gut-brain axis is now considered a legitimate therapeutic target for cognitive aging.
## How to Optimize Your Microbiome for Longevity
The good news is that the gut microbiome is highly responsive to lifestyle interventions, particularly diet. The following strategies have the strongest evidence for promoting a longevity-associated microbiome:
**Increase dietary fiber diversity.** The single most important dietary change for microbiome health is increasing the diversity and quantity of plant-based fiber. Different bacterial species ferment different types of fiber, so diversity of fiber sources — vegetables, fruits, legumes, whole grains, nuts, and seeds — promotes microbial diversity. The American Gut Project found that people who ate 30 or more different plant foods per week had significantly more diverse microbiomes than those who ate 10 or fewer.
**Consume fermented foods.** A 2021 randomized controlled trial in *Cell* found that a high-fermented-food diet (yogurt, kefir, fermented vegetables, kombucha) significantly increased microbiome diversity and reduced inflammatory markers over 10 weeks — effects that were not seen with a high-fiber diet alone. Fermented foods introduce live microorganisms and their metabolites directly into the gut.
**Limit ultra-processed foods.** Ultra-processed foods — which are low in fiber, high in emulsifiers, artificial sweeteners, and refined sugars — consistently reduce microbiome diversity and promote dysbiosis. Emulsifiers such as carboxymethylcellulose and polysorbate-80 have been shown to disrupt the intestinal mucus layer and promote inflammation in animal models.
**Exercise regularly.** Exercise independently promotes microbiome diversity and increases the abundance of SCFA-producing bacteria, independent of diet. Elite athletes have significantly more diverse microbiomes than sedentary individuals, and even moderate exercise (150 minutes per week of moderate-intensity activity) produces measurable microbiome benefits.
**Consider targeted probiotics and prebiotics.** Specific probiotic strains — particularly *Lactobacillus rhamnosus*, *Bifidobacterium longum*, and *Akkermansia muciniphila* — have demonstrated benefits for intestinal barrier integrity, inflammation, and metabolic health in clinical trials. Prebiotics (non-digestible fibers that selectively feed beneficial bacteria) such as inulin, fructooligosaccharides (FOS), and resistant starch can support the growth of beneficial species.
**Minimize unnecessary antibiotic use.** Antibiotics cause profound and sometimes long-lasting disruptions to the microbiome. While antibiotics are essential for treating bacterial infections, unnecessary use — particularly broad-spectrum antibiotics — should be avoided. When antibiotics are necessary, probiotic supplementation during and after the course can help restore microbiome diversity.
## The Future: Microbiome-Targeted Longevity Therapies
The field of microbiome-targeted longevity therapy is advancing rapidly. Fecal microbiota transplantation (FMT) — the transfer of gut microbiota from a healthy donor to a recipient — has already demonstrated efficacy for recurrent *Clostridioides difficile* infection and is being investigated for aging-related conditions. The concept of "young microbiome transplants" — transferring the microbiome of young, healthy donors to older recipients — is being explored in both animal models and early human studies.
More targeted approaches include the development of live biotherapeutic products (LBPs) — precisely defined microbial consortia designed to restore specific beneficial species — and postbiotics (purified microbial metabolites such as urolithin A or butyrate) that deliver the benefits of a healthy microbiome without requiring the microorganisms themselves.
The gut microbiome is not merely a reflection of health — it is an active participant in determining how long and how well we live. Tending to it with the same care we give to exercise, sleep, and nutrition may be one of the most impactful investments in longevity available to us today.
Written by
James Thornton, RD
Health & wellness expert contributing evidence-based content to Health Hub.